 The discovery, which has previously only be theoretically-predicted by scientists, helps explain the interactions between single oxygen molecules and titanium dioxide, giving the research teams a better idea of how the compound functions.
The discovery, which has previously only be theoretically-predicted by scientists, helps explain the interactions between single oxygen molecules and titanium dioxide, giving the research teams a better idea of how the compound functions.
The scientists also say that it could point to further enhancement of the compound, as well as different functions, including the possibility of combining CO2, titanium dioxide and light to produce fuel.
Studying the surface determines the application
“The surfaces of materials pose interesting fundamental questions, but are also important for applications”, said Ulrike Diebold, a physicist from the Institute of Applied Physics at TU Vienna.
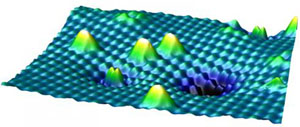
The researchers in Vienna were able to demonstrate how titanium dioxide interacts with air, showing how it interacts at an atomic level using a scanning tunneling microscope.
Specific to this type of microscope, the team employed a fine metal tip that was held close to the surface, while a voltage was applied to the tip to create what is called a tunneling current, which is measured and displayed as an image.
Microscopic images and a voltage determine atomic activity
The method produced images of the single atoms in the titanium dioxide, from which the team was able to locate vacancies in the atomic structure, enabling them to determine how differently ionized oxygen molecules become embedded on the surface of the compound.
“Our results clearly show how important these oxygen vacancies are for the chemical properties of titanium oxide”, stated Diebold.
“We were also able to show that we can alter the charge state of the photocatalytically active oxygen atoms. Perhaps in future it will be possible to produce more active oxygen-rich photocatalysts. These could be used to convert CO2 into useful hydrocarbons, with the help of the titanium dioxide and light.”